Each year, more than 8.2 million people die of cancer worldwide, and it is estimated that around 38.4% of people will be diagnosed with cancer at some point in their lives. Many of us have been impacted by cancer in some way, and the heartbreaking impact of cancer is only increasing as scientists predict that the incidence of cancer will nearly double by 2030, from 14.1 million new cases in 2020 to 23.6 million in 2030.
Naturally, in the field of medicine, curing cancer is one of the major pursuits of doctors, scientists, researchers, and more. Throughout history, medical professionals have developed treatments such as surgery and chemotherapy that have saved the lives of many cancer patients, but recently, new treatments have arose, and many of these treatments are revolutionizing medicine and changing the lives of millions around the world.
What really is cancer?
The word “cancer” came from the Latin word for “crab,” since physicians believed that cancer tumors looked similar to a crab. Fossils and mummies also show evidence of cancer, which proves that cancer has existed throughout history.
However, what exactly is cancer? Normally, cells divide and self-destruct in a controlled and orderly manner, but cancer cells are abnormal cells that divide uncontrollably and can spread to other tissues and push out normal cells. It can be caused by a number of factors, including genetics and the environment.
What causes cancer?
One main cause of cancer is mutations, which is when errors happen during cell division. Normally, daughter cells are identical to the parent cell, but sometimes errors can occur causing mutations. Often times, these mutations can be dangerous, causing cancer or other diseases.
There are two types of mutations, hereditary mutations and somatic mutations. Hereditary mutations are inherited and can be passed down through generations, as the mutation is in every cell in your body, including germ cells, which are sperm and egg cells. Somatic mutations are acquired mutations that happen due to environmental factors, such as radiation, or errors in cell division. The mutations that occur in somatic cells cannot be passed down to offspring, since they only occur in certain cells, not all of the cells in the body.
However, having a cancer mutation does not necessarily mean you will have cancer. In fact, environmental factors play into a patient’s risk of developing cancer, although different people will require different environmental impacts to develop cancer. For example, two people could be exposed to the same amounts of the same carcinogen, but only one develops cancer.
What is the two-hit hypothesis?
One hypothesis that explains this is Dr. Alfred G. Knudson Jr.’s Two-Hit Hypothesis, which states that people who have inherited one mutated allele, or “hit”, on their tumor-suppressor gene have an increased risk for cancer because they only require another “hit,” possibly caused by environmental factors, to develop cancer. Meanwhile, people with normal alleles will require two “hits” caused by environmental factors to develop cancer, making it much more likely for someone who inherited a mutated tumor-suppressor allele to develop cancer.
“…I consider that there are three categories of tumors. One is the embryonal tumor. It arises in tissues where the stem cells are multiplying. So, there are many rounds of cell division before differentiation occurs. I made calculations on whether people who inherit one mutation in the germline could develop a second mutation, or hit, based on the usual rate of somatic mutations. I found that they could, that the ordinary mutation rates suffice for a second hit to occur. But I also calculated that people who do not inherit a germline mutation can develop a clone of cells that have one somatic mutation. Rarely, one of these cells may develop a second mutation and become a tumor. In fact, I have estimated that almost 30% of us have clones of oncehit cells in our retina, but differentiation occurred before a second hit could occur. I estimated that the second, cancercausing hit happens to only 1 in 30,000 people.”
Dr. Alfred Knudson on mutations that drive tumor development
What are some common treatments for cancer?
While distinct cancers are treated differently, the most common treatment for cancer is surgery, chemotherapy, or radiation. Some of these treatments are non-specific and attack all types of growing cells, including hair cells, gut cells, and immune cells, which causes significant side effects such as hair loss, infection, nausea, and vomiting. However, recent discoveries in cancer treatments have given rise to the use of immunotherapy, a treatment that uses a patient’s own immune system fight cancer cells.
The history of immunotherapy
For thousands of years, doctors noticed that tumors occasionally shrunk or disappeared after infections, but the first doctors to try to use the immune system to cure cancer were German physicians Fehleisen and Busch, who noticed the tumor shrinking after an erysipelas infection.
Typically, cancer cells can keep the immune system from recognizing or destroying the cancer through genetic changes, surface proteins, or changing the surrounding cells so the patient’s immune system doesn’t recognize that the cancer cell is a dangerous cell that needs to be killed.
However, in 1891, Dr. William Bradley Coley shrunk or cured over 1,000 patients by injecting them with live or inactivated Streptococcus pyogenes and Serratia marcescens, which became the first recorded immune treatment for cancer. Yet, Coley’s discoveries were widely forgotten, until 1945, when many new discoveries in immunology and oncology led scientists to revisit the possibility of immunotherapy.
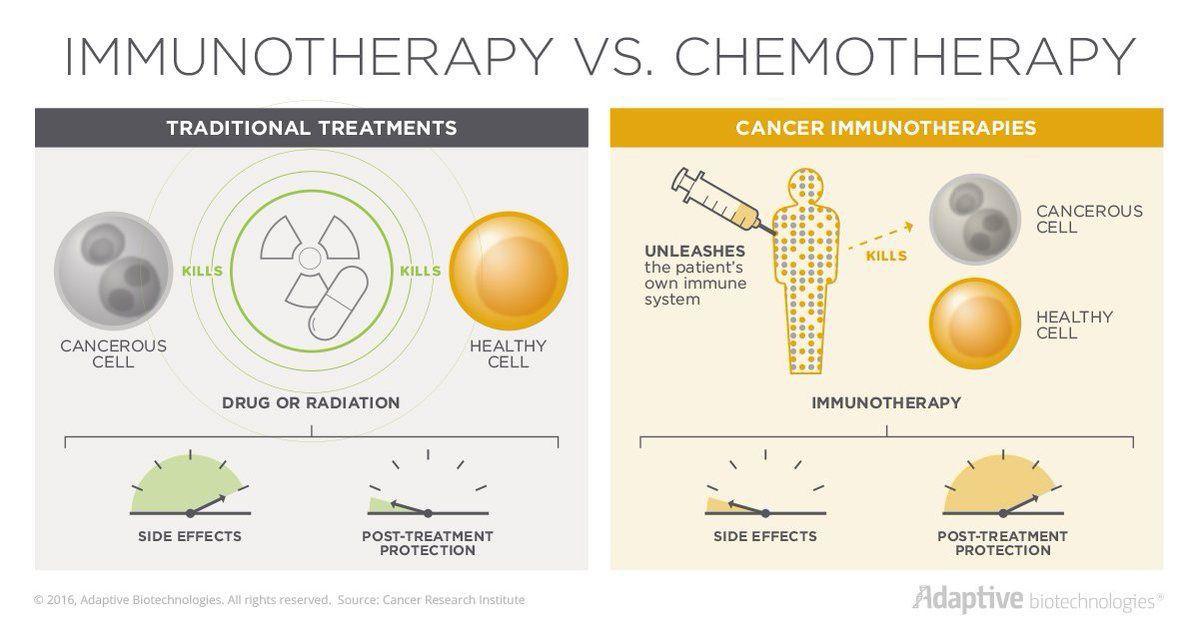
How does immunotherapy work?
Today, immunotherapy has become another form of treatment, along with surgery, chemotherapy, and radiation, and it is most commonly used to treat bladder cancer, breast cancer, colon cancer, and more.
One type of immunotherapy includes immune checkpoint inhibitors, which uses drugs to block immune checkpoints that typically control immune responses from getting too strong. This allows the immune system to unleash a strong response to cancer cells, possibly killing the cancer.
Another type of immunotherapy is T-cell transfer therapy, which takes immune cells from a patient’s tumor, selects for the best immune cells that attack the cancer or genetically modified a patient’s T-cells in a laboratory, grows the best cells in large amounts, then are injected back into the patient to identify and attack the cancer.
In addition, monoclonal, or therapeutic, antibodies work in a somewhat similar way, as these antibodies are genetically modified in a laboratory to attach to certain targets on a cancer cell that will mark the cell for destruction by the immune system. Most immunotherapy agents are given through intravenous therapy (IV), oral pills or drugs, topical creams, or intravesical therapy that goes into the patient’s bladder.
What is CAR T-cell therapy?
One of the main types of T-cell transfer therapy is CAR T-cell therapy, which stands for chimeric antigen receptor, and the process produces special receptors that allow the T-cells to bind to specific antigens or proteins on the surface of a tumor.
First, a patient’s blood is drawn, and the T-cells are separated out. These T-cells are then genetically modified using disarmed viruses to produce a specific receptors, and the modified T-cells will be grown in large batches.
Finally, the modified T-cells will be infused into the patient after a process called lymphodepleting chemotherapy, which gets rid of current T-cells, and this process should allow patients to have modified T-cells that will continue to grow in their body and attack tumors.
CAR T-cell therapy has showed promising results, as 90% of the acute lymphocytic leukemia (ALL) patients treated with CD-19-targeted CAR T-cell therapy showed no signs of cancer after the treatment. CAR T-cell therapy is becoming a widely used therapy for lymphomas, which is important as often times lymphomas and leukemias often do not have tumors making it difficult to treat with surgery and other standard methods.
What are the side effects of CAR T-cell therapy?
However, as with most disease treatments, there are risks and side effects to CAR T-cell therapy. The most common side effect of this therapy is cytokine release syndrome (CRS), which releases huge amounts of cytokines, the chemical messengers used by the immune system to communicate, causing symptoms such as low blood pressure and high fevers.
Other side effects include B-cell aplasia, which causes B-cells to die off in large numbers, cerebral edema, which is swelling of the brain, and neurotoxicities, but most of these effects are reversible or can be resolved using supportive therapy.
What might CAR T-cell therapy be used for in the future?
Research in CAR T-cell therapy has been rapidly progressing, and with high success rate in lymphomas and other blood cancers, scientists are looking to use this therapy on solid cancers. However, while using CAR T-cell therapy for solid cancers isn’t showing the same success rate, scientists believe that other similar types of therapies are promising treatments for patients suffering from cancer.
In conclusion…
For thousands of years, physicians and scientists have searched for ways to cure cancer, and today, new treatments and discoveries, including immunotherapy, have helped save millions of lives. Through clinical trials and research, scientists have found that immunotherapy is a promising treatment for cancer, specifically lymphomas and leukemias, which were harder using standard methods to treat due to the lack of tumors in some of these cancers. Cancer treatments have gone a long way, and as medical discoveries continue to pave the way for mankind, and maybe one day, we’ll be able to say, “it’s just cancer”.
*Disclaimer: This blog is not intended to provide health or medical advice, please seek help from a licensed physician if you have any specific health questions or concerns!*
Bibliography
Cancer Statistics. 27 Apr. 2018, www.cancer.gov/about-cancer/understanding/statistics.
“Early History of Cancer.” American Cancer Society, American Cancer Society, 4 Jan. 2018, www.cancer.org/cancer/cancer-basics/history-of-cancer/what-is-cancer.html.
“What Is a Gene Mutation and How Do Mutations Occur?” U.S. National Library of Medicine, National Institutes of Health, 17 Aug. 2020, ghr.nlm.nih.gov/primer/mutationsanddisorders/genemutation.
Knudson’s “Two-Hit” Theory of Cancer Causation, 12 Feb. 2018, www.foxchase.org/about-us/history/discoveries-fox-chase-research/knudsons-two-hit-theory-cancer-causation.
Dobosz, Paula, and Tomasz Dzieciątkowski. “The Intriguing History of Cancer Immunotherapy.” Frontiers in immunology vol. 10 2965. 17 Dec. 2019, doi:10.3389/fimmu.2019.02965
“What Is Cancer?” American Cancer Society, 8 Dec. 2015, www.cancer.org/cancer/cancer-basics/what-is-cancer.html.
“What Types of Cancer Can Be Treated With Immunotherapy?” Edited by Osama Rahma, Dana-Farber Cancer Institute, Dana-Farber Cancer Institute, 21 Apr. 2020, blog.dana-farber.org/insight/2020/04/what-types-of-cancer-can-be-treated-with-immunotherapy/.
“What Is Cancer?” National Cancer Institute, 9 Feb. 2015, www.cancer.gov/about-cancer/understanding/what-is-cancer.
“CAR T Cells: Engineering Immune Cells to Treat Cancer.” National Cancer Institute, 30 July 2019, www.cancer.gov/about-cancer/treatment/research/car-t-cells.
“Immunotherapy to Treat Cancer.” National Cancer Institute, 24 Sept. 2019, www.cancer.gov/about-cancer/treatment/types/immunotherapy.
Simon, Stacy. “Facts & Figures 2019: US Cancer Death Rate Has Dropped 27% in 25 Years.” American Cancer Society, American Cancer Society, 8 Jan. 2019, www.cancer.org/latest-news/facts-and-figures-2019.html.


